Spotlights
Tiêu đề tương tự
Nhà thiết kế sản phẩm y tế, Nhà thiết kế thiết bị y sinh, Nhà thiết kế thiết bị chăm sóc sức khỏe, Nhà thiết kế thiết bị y tế, Nhà thiết kế công nghệ y tế, Nhà thiết kế thiết bị lâm sàng, Kỹ sư thiết bị y tế, Chuyên gia phát triển thiết bị y tế, Nhà thiết kế đổi mới chăm sóc sức khỏe, Kỹ sư thiết kế thiết bị y tế
Mô tả công việc
Nhà thiết kế thiết bị y tế và sức khỏe chịu trách nhiệm thiết kế và phát triển các thiết bị và thiết bị y tế sáng tạo và thân thiện với người dùng. Họ kết hợp chuyên môn của họ trong các nguyên tắc thiết kế, khái niệm kỹ thuật và yêu cầu chăm sóc sức khỏe để tạo ra các thiết bị an toàn, hiệu quả và khả thi về mặt thương mại.
Trách nhiệm công việc
- Khái niệm hóa và thiết kế: Phối hợp với các nhóm chức năng chéo, bao gồm các kỹ sư, bác sĩ lâm sàng và quản lý sản phẩm, để động não và phát triển các khái niệm thiết kế cho các thiết bị y tế mới hoặc cải tiến các thiết bị hiện có.
- Nghiên cứu và phân tích: Tiến hành nghiên cứu kỹ lưỡng về nhu cầu của người dùng, xu hướng thị trường và tiến bộ công nghệ trong ngành chăm sóc sức khỏe để thông báo cho quá trình thiết kế. Phân tích và giải thích phản hồi của người dùng, dữ liệu lâm sàng và các yêu cầu quy định để đảm bảo tuân thủ và khả năng sử dụng.
- Phát triển nguyên mẫu: Tạo nguyên mẫu của các thiết bị y tế bằng nhiều công cụ và kỹ thuật khác nhau, chẳng hạn như thiết kế hỗ trợ máy tính (CAD), tạo mẫu nhanh và in 3D. Kiểm tra và đánh giá các nguyên mẫu để đánh giá chức năng, công thái học và an toàn.
- Tối ưu hóa thiết kế: Phối hợp với các nhóm kỹ thuật để tinh chỉnh và tối ưu hóa thiết kế thiết bị dựa trên tính khả thi kỹ thuật, khả năng sản xuất và hiệu quả chi phí. Tiến hành lặp lại thiết kế và nghiên cứu khả thi để đảm bảo tính tương thích với các quy trình sản xuất.
- Tài liệu và Tuân thủ: Tạo tài liệu thiết kế chi tiết, bao gồm bản vẽ kỹ thuật, thông số kỹ thuật và kế hoạch xác minh thiết kế. Đảm bảo tuân thủ các tiêu chuẩn quy định có liên quan, chẳng hạn như quy định của FDA, tiêu chuẩn ISO và chỉ thị về thiết bị y tế.
Các kỹ năng cần thiết trong công việc
- Thành thạo phần mềm CAD và các công cụ thiết kế khác (ví dụ: SolidWorks, AutoCAD, Adobe Creative Suite).
- Hiểu biết sâu sắc về các nguyên tắc thiết kế, kỹ thuật yếu tố con người và các nguyên tắc khả năng sử dụng trong bối cảnh chăm sóc sức khỏe.
- Kiến thức về các quy định và tiêu chuẩn về thiết bị y tế (ví dụ: quy định của FDA, ISO 13485, IEC 60601).
- Làm quen với các quy trình sản xuất và cân nhắc cho sản xuất thiết bị y tế.
- Kỹ năng giải quyết vấn đề và tư duy phê phán tuyệt vời để vượt qua các thách thức thiết kế và tối ưu hóa chức năng của thiết bị.
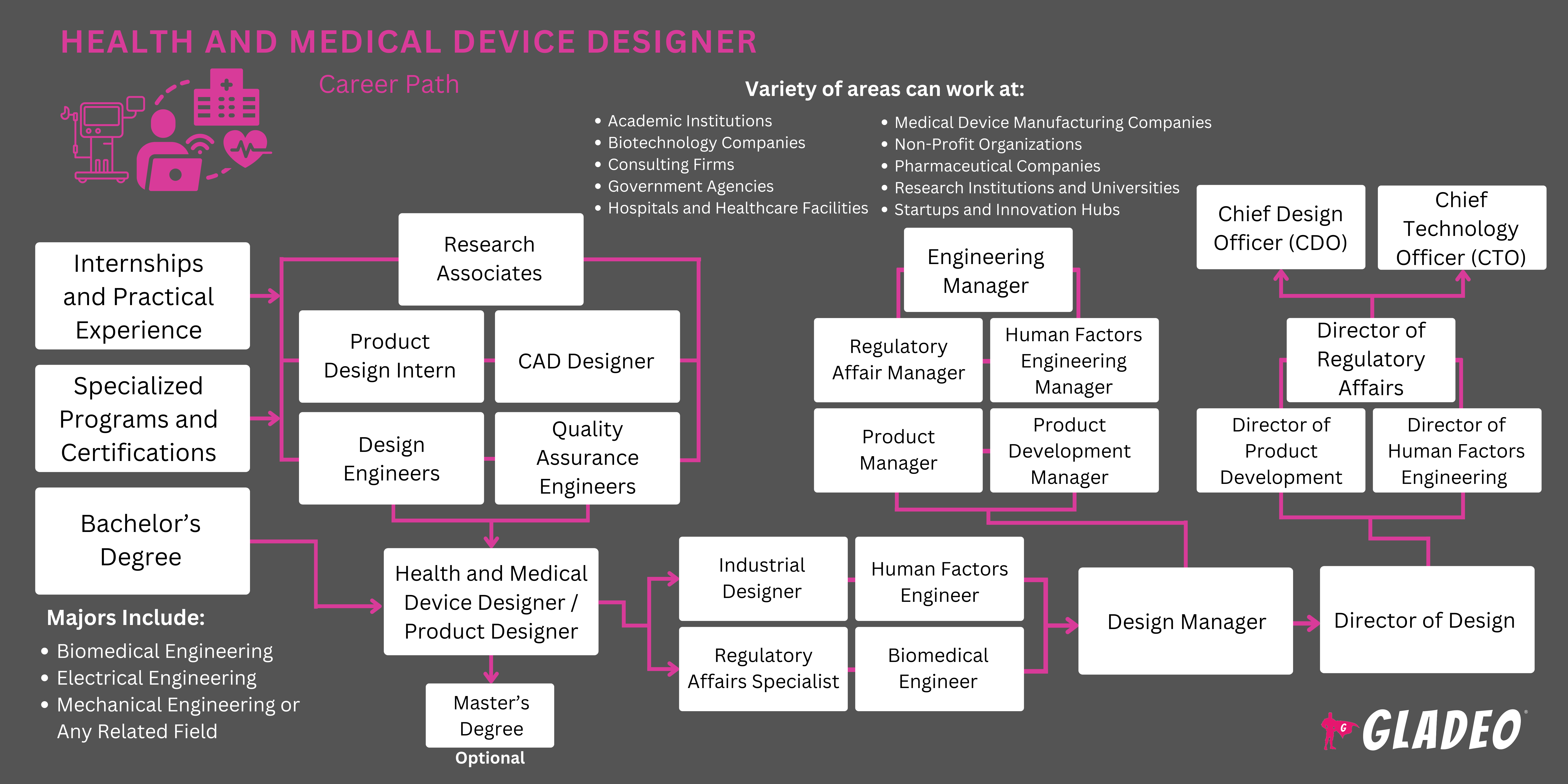
Lộ trình điển hình
Nguồn cấp tin tức

Việc làm nổi bật

Các khóa học và công cụ trực tuyến









